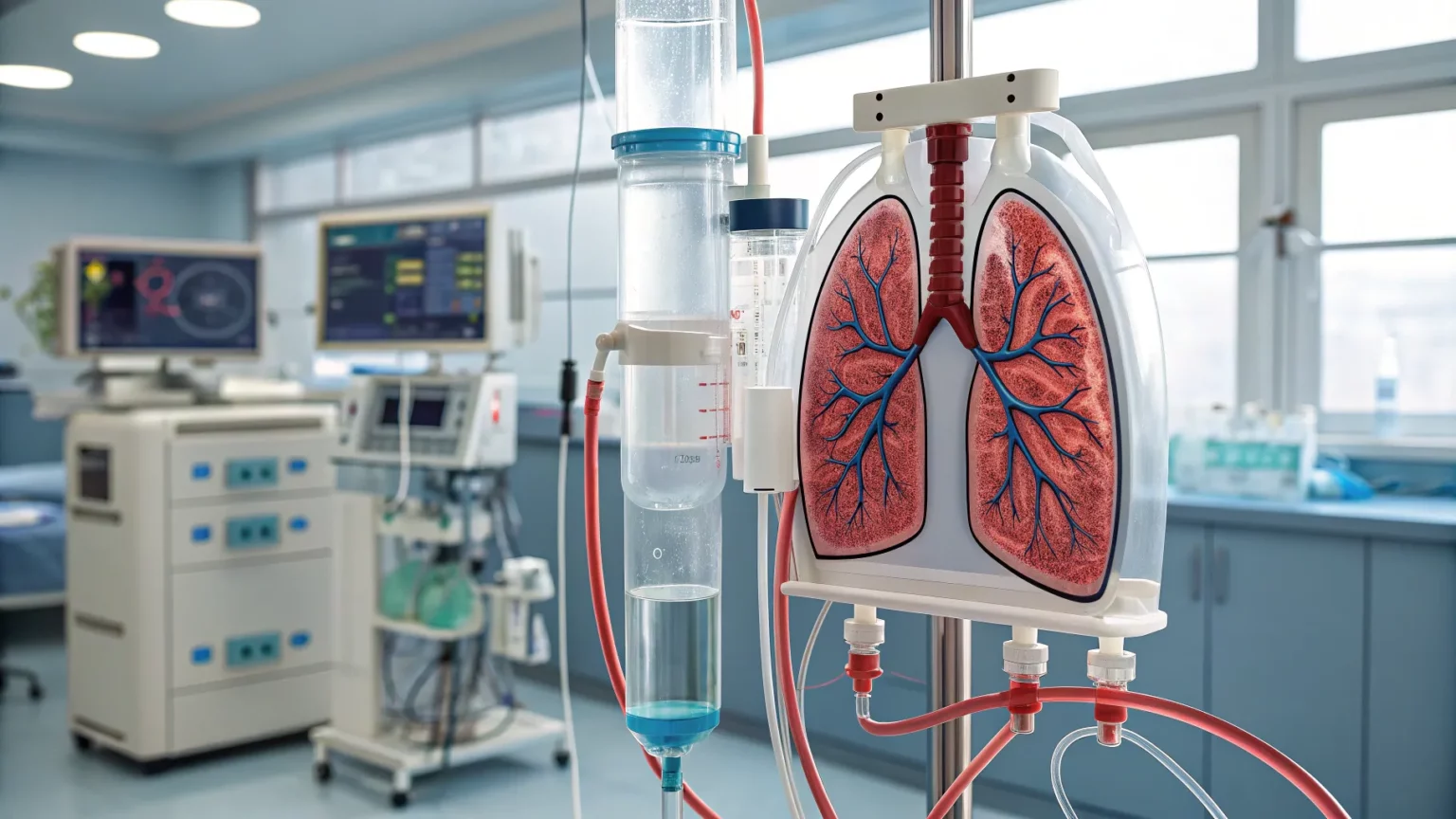
A prototype artificial lung system could keep people alive for weeks without natural lungs, according to a short demonstration shared by its developers. The device uses tubes and pumps to oxygenate blood and maintain blood flow, echoing the core function of human lungs. The effort signals a push to buy time for patients with severe respiratory failure, especially those awaiting transplant or recovering from catastrophic illness.
The system was shown operating outside the body, acting as a temporary substitute for a failing organ. It aims to stabilize patients long enough for other treatments to work. The announcement did not include a release date, clinical site, or specific trial results.
How the Device Works
“A new artificial lung system might keep people without lungs alive for weeks. Like real lungs, tubes and pumps oxygenate blood and maintain blood flow.”
The description matches the principles of extracorporeal support. Blood is drawn from the body, passed through a membrane where oxygen is added and carbon dioxide is removed, and then returned to circulation. Pumps drive flow to match the patient’s needs. Safety controls are required to prevent clots, bleeding, and infection.
Developers suggest the unit could function for weeks, which is a key threshold in intensive care. That duration could bridge patients through acute illness or a waitlist period.
Background: From ICU Support to Long-Term Bridge
Hospitals already use extracorporeal membrane oxygenation, or ECMO, for severe lung failure. ECMO can sustain gas exchange when ventilators are not enough. It is complex, labor intensive, and carries risks. Typical runs can last days to weeks, depending on the case and center expertise.
The new system appears to build on these lessons, aiming for a more durable and perhaps more mobile setup. Fewer complications and easier management would mark progress. Clear evidence from animal studies or early human use will be needed to confirm the claims.
Who Could Benefit
Several groups could gain if the device performs as described:
- Patients with acute respiratory distress syndrome after infection or injury.
- People with lung failure awaiting transplant organs.
- Patients recovering from toxic exposures or trauma where lungs need time to heal.
For transplant centers, a weeks-long bridge could reduce deaths on the waitlist. For community hospitals, transfer protocols would be crucial to ensure timely access.
Clinical Questions and Safety Risks
Experts will watch for data on clotting, bleeding, and hemolysis. Blood contacting artificial surfaces can trigger inflammation and clots. Anticoagulation reduces clotting but raises bleeding risk, especially after surgery or in trauma.
Infection control is another challenge. Indwelling cannulas create entry points for bacteria. Teams must manage sterile technique and monitor for sepsis. Device durability, membrane performance over time, and pump reliability also matter.
Mobility is a frontier for extracorporeal support. Allowing patients to sit, stand, and do therapy can improve outcomes. Any new system should show how it supports movement without compromising safety.
What Evidence Is Still Needed
Developers did not present trial data with the announcement. Independent testing will be needed to assess outcomes such as survival, complication rates, and quality of life. Comparisons with current ECMO protocols would help hospitals judge value.
Key measures to watch include:
- Days supported without major bleeding or thrombosis.
- Rates of stroke, infection, and device failure.
- Successful bridging to transplant or recovery.
- Nurse and perfusionist workload and training demands.
Ethics, Access, and Cost
Extended life support raises ethical questions when the path to recovery is uncertain. Clear criteria for starting and stopping support are essential. Families need transparent goals and timelines.
Cost will shape adoption. Extracorporeal care is resource heavy and requires trained staff. If the device reduces complications or staffing time, hospitals could see savings. If it adds complexity, access may cluster in large centers.
Outlook for Patients and Hospitals
If confirmed, weeks-long lung replacement outside the body could mark a practical step for respiratory care. It would not cure underlying disease, but it could buy time for healing or transplant. For now, the promise remains tied to pending data.
Hospitals and patients should watch for peer-reviewed results, regulatory review, and details on training. Clear protocols, strong safety records, and manageable costs will determine whether this system reaches everyday care.
The early message is simple yet ambitious: mechanical lungs may soon keep people alive long enough for medicine to do the rest.